|
Then we derive the rest of our favorites, the watt, the volt, and the ohm. Next up after the ampere comes the coulomb and charge on an electron. It is an SI base unit, the only electrical unit derived from the outcome of an experiment. 
This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Henry Agnew, and Lance S. The standard electrical units are defined in a specific order. The atomic mass unit (amu) is a unit of mass equal to one-twelfth the mass of a carbon-12 atom.The trends for the entire periodic table can be seen in Figure 8.4. This trend is illustrated for the covalent radii of the halogens in Table 8.4. As a result, a neutral atom must have an equal number of protons and electrons. Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. The positive charge on a proton is equal in magnitude to the negative charge on an electron.Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron).Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).Protons are bound together in an atom's nucleus as a result of the strong nuclear force. A coulomb is an enormous charge - two 1 C charges that are 1 m apart exert a force of 9 x 109 newtons (. It is the reflected longitudinal displacement of a granule from. Protons are a type of subatomic particle with a positive charge. Conversely, the charge of an electron is -1.6 x 10-19 C. The elementary charge is the electric charge carried by a single electron or proton.Electrons are a type of subatomic particle with a negative charge. 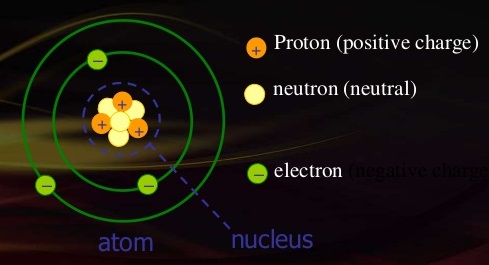
When we write the symbol for an atom, we can place its mass number at the top left and its atomic number at the. \): Properties of Subatomic Particles Particle Electric charge is the physical property of matter that causes it to experience a force when placed in an electromagnetic field. has no overall charge, because it has an equal number of protons and electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
